Connecting the C4D to the Beckman Coulter CE
This application note describes how to connect the eDAQ C4D system to the P/ACE MDQ and PA 800 plus capillary electrophoresis instruments from Beckman Coulter.
Contents
Introduction
The eDAQ C4D can be connected to the Beckman Coulter CE instruments, providing contactless conductivity detection for the instrument. Both the C4D headstage and UV detector can be connected to the capillary at the same time, enabling both types of detection to be performed with a single sample injection.
Equipment Required
- C4D hardware unit, either:
- ER225 C4D Data System to record both C4D and UV signals in the eDAQ software.
- ER815 C4D Detector to record both C4D and UV signals in Beckman Coulter software (may require an external A/D interface if the CE doesn't have an analog input),
- ER815R C4D Detector to record only the C4D signal in PowerChrom, or
- ER825R Two Channel C4D Detector to record both C4D and UV signals in PowerChrom or Beckman Coulter software
- ET120 C4D Headstage for Capillary Electrophoresis
- Beckman Coulter CE instrument
- Two sets of the External Detector Adapter (EDA) part number 144822, which can be purchased from Beckman Coulter.
- Signal cable:
- For ER225: EC008 BNC to alligator clips cable (with alligator clips removed), or
- For ER815: BNC to BNC Cable
- Trigger cable:
- For ER225: use a simple tinned end to tinned end double-wire cable, or
- For ER815: EC073 Trigger cable
- EC020 Standard Test Solutions for C4D Applications
Choice of Software
The data from both the eDAQ C4D and Beckman Coulter UV/LIF/PDA detectors can be displayed in real time, using either the eDAQ software or the Beckman Coulter PA 800 plus Software. Use the ET120 C4D Headstage and select either the:
- ER815 C4D Detector can be configured by the user to use either PowerChrom or third-party software like Beckman Coulter software. The analog output from the C4D may be connected to the CE, so the PA 800 plus Software records data from both detectors. This may require an external A/D interface if the CE doesn't have an analog input. The delta K signal from the C4D Detector should be connected to the A/D interface input. The A/D interface is then connected to the computer using a serial connection. The ER825R model includes PowerChrom software and can record the signal from both C4D and UV detectors.
- ER825R Two Channel C4D Detector, can also be configured by the user to use either PowerChrom or third-party software like Beckman Coulter software. The ER825R model includes PowerChrom software and can record the signal from both C4D and UV/LIF/PDA detectors.
- ER225 C4D Data System to use the eDAQ software. The analog output from the UV/LIF/PDA detector (on the CE) may be connected to the eDAQ system, so the eDAQ software records data from both detectors.
Analog Outputs on the CE
There are three BNC analog outputs located on the left side panel of the CE instrument, as shown in Figure 3. The outputs are 1000 mV full scale deflection. For PA 800 plus firmware version 1.5 or higher, the output is programmable from the control software. In version 1.2, 1000 mV is equal to 1 absorbance unit (AU) with a fixed 10% negative offset. The high voltage output is the uncontrolled parameter, current or voltage, of the high voltage power supply. In this instance, 1000 mV is equal to 30,000 V or 300 µA in all firmware versions.
External Detector Adapter
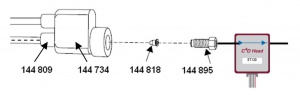
The Beckman Coulter CE instrument uses a liquid to cool the capillary. The eDAQ ET120 C4D Headstage can be connected to the capillary using two sets of the External Detector Adapter (EDA) part number 144822, which can be purchased from Beckman Coulter. Each EDA kit includes:
- 144734 loop reservoir
- 144895 red screw
- 144818 red double end conical gasket
- 144834 which includes:
- 970298 (x4) black O-ring
- 966233 (x4) blue conical gasket
- 144809 (x4) blue screw
- 144832 tubing
The assembled EDA is shown in Figure 4. The continuous capillary has been labelled as three parts. Part 3 should be as short as possible.
The length of Part 1 should be the same length as the overall length of the capillary when the UV detector is used. This allows for the same separation length. The liquid loop should be as short as possible to save on cooling liquid and space in the cartridge. The tubing may be bent while immersing in hot water and then placed in cold water to preserve the shape.
The C4D cable may exit the cartridge and instrument though grooves provided.
The connections of the capillary with the screws and gaskets is shown in Figure 5.
Ensure that you position the C4D headstage near the end of the capillary that is grounded, away from the end of the capillary where the high voltage is applied. This is to ensure that the high voltage doesn’t arc from the background electrolyte inside the capillary, through the thin wall of the capillary, to the headstage and on to the C4D hardware, which could damage the equipment.
Triggering Start of Recording
The trigger signal (found on the left side of the CE instrument) may be used to trigger the eDAQ software to start recording. Using a cable, connect pin 9 (CE instrument) to the TRIG + (C4D hardware unit) and pin 10 to TRIG —, as shown in Figure 6. Setting up of software:
- PA 800 plus Software: make a Method, go to Instrument Setup, then in Time program, enter Time = 0 and Event = Relay 2 On.
- eDAQ PowerChrom software: In the Inject Settings window, select "Wait for Inject". Or in eDAQ Chart software: In the menu Setup, Trigger, choose External Trigger to start data recording.
Citations
- The eDAQ C4D was used with the P/ACE MDQ instrument (Beckman Coulter) for the simultaneous determination of kanamycin, its related substances and sulphate in kanamycin sulphate. Kanamycin is an aminoglycoside antibiotic that lacks a strong UV-absorbing chromophore, so direct UV detection cannot be used. The limit of detection (LOD) and limit of quantitation (LOQ) for kanamycin were 0.7 and 2.3mg/L, respectively. DOI: 10.1002/jssc.201100267
- The quantification of amikacin in bronchial epithelial lining fluid in neonates was performed using the Beckman Coulter P/ACE MDQ and the eDAQ C4D. DOI: 10.1128/AAC.00277-11
- The P/ACE MDQ was used with the eDAQ C4D, collecting the C4D data in the Beckman Coulter 32 Karat software, for the analysis of amikacin and its related substances. Amikacin is a semisynthetic aminoglycoside antibiotic that lacks a strong UV absorbing chromophore or fluorophore. The LOD and LOQ were 0.5 mg/L and 1.7 mg/L, respectively. The method yielded an effective improvement in simplicity and analysis time (6 minutes). This is important for routine application compared to the method obtained using UV detection after derivatization (35 minutes), and to the liquid chromatography method (75 minutes). DOI: 10.1002/elps.201100688
- El-Attug et al used the eDAQ C4D and Beckman Coulter CE for the detection of tobramycin and its related substances. The LOD and LOQ are 0.4 and 1.3 mg/L respectively. It was noted that contactless conductivity detection is useful for the analysis and assay of aminoglycosides such as tobramycin, and that it can be a good alternative to derivatization in case of non UV-absorbing substances. DOI:10.1016/j.jpba.2011.09.032