Procedure for Microchip Electrophoresis with C4D with the ET121
Contents
Equipment
- ER225 C4D Data System
- ET121 Microchip Platform with C4D Electrodes
- ER430 High Voltage Sequencer
- EC230 High Voltage Cables
- Computer with PowerChrom and QuadSequencer softwares
- EC020 Standard Test Solutions:
- BGE = 0.5M acetic acid
- Sample = 1mM LiCl, KNO3 and Na2SO4 in deionised water
- EC020 Standard Test Solutions:
- Chips
- start with ChipShop 02-0772-0202-05, Zeonor material (a type of Cyclic Olefin Copolymer (COC)), with 100 µm base
- then use custom made chip with 60 µm base (might be similar to 02-0772-0202-05)
- the channel has a square cross section (not circular); Main channel width, depth, length= 75 µm, 75 µm, 87 mm; 0.49 µL main channel volume; dimensions of chip File:1407 chip for rereleased ET121.pdf (68 KB PDF)
- One 20 - 200 µL pipette, with pipette tips
- One syringe 5 mL
- Deionised water
- Lint-free tissues
Preparing the Hardware
The Microchip Electrophoresis Kit contains many components and cables. Please follow the instructions below in the order listed.
- Check you have all the components listed in the packing lists.
- Install the QuadSequencer software and PowerChrom software on the computer. Ensure you have installed the latest versions of softwares from https://www.edaq.com/software-downloads
- Connect the High Voltage Sequencer (HVS) to the computer with a USB cable, as described in the instruction manual. Connect the power cable. Do not turn it on yet.
- Connect the C4D Data System to the computer with a USB cable, as described in the instruction manual. Connect the power cable. Do not turn it on yet.
- Connect the Chip Platform to the front of the C4D Data System.
- Connect the coloured EC230 High Voltage Cables to the HVS as follows: blue cable to Output 1, red cable to Output 2, yellow cable to Output 3, and black cable to Output 4.
- You will need to create an interlock system for the HVS. Connect the interlock system between between the Chip Platform and the HVS. Warning: the interlock is designed to disarm the HVS if the interlock is open. Do not try to bypass the interlock.
- Grounding is important. Use a green ground cable to connect the green connector at the back of the C4D Data System, to the green connector at the back of the HVS.
- The HVS can trigger the PowerChrom software to start recording. Use the red and black EC201 trigger cable to connect the “CTL1 +” at the back of the HVS to the “TRIG +” at the back of the C4D Data System. Connect the HVS “CTL1 –” to the C4D “TRIG –”. Make sure you push the connection port block fully into each hardware unit. See the video (The software screen shots may look a bit different).
- Turn on the HVS. The driver will be installed if this is the first use.
- Open the QuadSequencer software. Move and resize the window so it occupies the left half of the screen.
- Turn on the C4D Data System. The driver will be installed if this is the first use.
- Open the PowerChrom software. Ensure the software has setup the hardware unit; you should see the Easy Access window, not the Hardware Unavailable window. Move and resize the PowerChrom software screen so it occupies the right half of the screen.
- Setup the trigger commands in the software packages as shown below:
- QuadSequencer software: in the menu Setup, Hardware, for the Digital Outputs, select Contact Closure. Also, in the Safety box, make sure "Float and I/O open at end/stop" is clicked.
- PowerChrom software: in the menu Edit, Preferences, Digital IO Settings, select External Trigger Mode as Voltage Level (TTL).
- PowerChrom software: click Manual Run to see the Manual Sampling window; click Inject Settings and select "Wait for Inject".
- Sequencer software: in the later step, when you are setting up the sequence, remember to select High/Closed, under Digital Output 1, during the separation step, as this will send the trigger command.
- Use gloves to place the empty microchip on the Chip Platform. See the note on Preparing the Chip. If you look at the chip closely, you may see the channel running along the chip. Place the channel above the white line on the platform. This will ensure that the channel sits on top of the C4D electrodes, which is essential for the C4D to detect analytes in the channel. Position the chip so that the reservoir at the end of the chip is about 1 cm away from the gold C4D electrodes.
- Secure the chip to the platform using the rectangular block with screws. Don't tighten the screws too much; it's just to stop the chip moving. Check the chip's reservoir is on top of the gold C4D electrodes. See the note on Positioning the Chip on the ET121 Platform.
- You should now dilute the sample (1mM LiCl, KNO3 and Na2SO4) to 100µM using deionised water.
How to Fill the Channel with BGE
Simply pipetting the background electrolyte (BGE) into reservoir of the ChipShop chip does NOT fill the main channel by capillary action! You must use syringe to push the BGE to fill main channel; this can be observed by looking at C4D signal and seeing a change in conductivity as the air is replaced with the BGE.
Here is a procedure for filling the channel with BGE:
- Have PowerChrom software running: In the PowerChrom software, in the Hardware Settings, C4D Amplifier, set the recording range to 5 V and apply 5 Hz low pass filter. Check the C4D settings are 80 % amplitude, 250 kHz frequency, headstage gain ON. Then zero the signal. Click OK and then Start.
- Pipette 30 µL of BGE in reservoir 4 (50 µL is too much as it will overflow when the pipette is pushed into reservoir luer); capillary action alone doesn’t fill channel.
- Place the syringe filled with BGE (make sure there is absolutely no air in the syringe or its luer neck) into reservoir 4 and push down gently so it makes seal with luer; this should fill the chip's channel with BGE, which can be observed by big change in C4D signal (about 300 mV change in voltage).
- Push on syringe while looking at reservoirs 1, 2 and 3 with magnifying glass; make sure you see BGE rising from the hole inside EACH reservoir; this ensures all the channels are filled, with no air bubbles.
- Continue to push on the syringe for one minute to flush the channel of the chip. You will have to continually remove the BGE from reservoirs 1, 2 and 3 as they are filled from underneath.
Check the Current is Flowing
To check there are no air bubbles and that the current will flow:
- Remove the BGE from all reservoirs.
- Pipette 50 µL of fresh BGE to each reservoir.
- Position the high voltage electrodes into each reservoir using the Lead Wire Clamp as shown in the photo. See the note on Positioning of the High Voltage Electrodes.
- Use a magnifying glass to check each electrode is immersed in liquid, but is NOT at the very bottom of the reservoir inside the hole leading to the chip's channel.
- Apply 500 V at reservoir 2 and ground at reservoir 4. First, arm the HVS by pushing the red button, on the front of the HVS, for three seconds. Then in the QuadSequencer software, click the Immediate tab. Type in the relevant voltages for each reservoir and click Apply. From now on, do not touch or approach the chip platform.
- Check there is a current flowing along the channel of the chip by looking at the current values displayed in the cross diagram of the QuadSequencer software. You may observe about 3.5 µA microamps of current flowing while applying 500 V along the chip' channel. The software should display about +3.5 µA on reservoir 2 and -3.5 µA on reservoir 4; don't worry if the currents don't match exactly. If the voltages are displayed in red text, the HVS is having difficulty applying the voltage; see the HVS manual.
- A flowing current shows there is no air gap. You should also see slight change in C4D signal (in PowerChrom software) when applying a voltage along the channel.
- Increase the current on reservoir 2 to 1000V to check that the current approximately doubles.
- Click Float Now, and then apply 500 V across reservoirs 1 and 3 (500V on reservoir 1 and Ground on reservoir 3) to check there is current flowing through these reservoirs as well. The current will be much higher, possibly around 40 µA, because the distance between these reservoirs is much smaller. Again, you can double the current to see the effect on the current.
- Check the currents are stable, not fluctuating.
- Click Float Now to stop the voltage being applied. You can also disarm the HVS by either clicking Disarm in the QuadSequencer software or briefly pushing the red button on the front of the HVS.
- Check to see if gas bubbles have formed next to the high voltage electrodes. See the note on Positioning of the High Voltage Electrodes and Gas Bubbles.
How to Run a Blank
Now you have prepared the experiment, filled the chip's channel with BGE, and checked that the current is flowing, you are ready to run a blank:
- In the PowerChrom software, in the Hardware Settings, C4D Amplifier, change the recording range to 100 mV and apply 5 Hz low pass filter. Check the C4D settings are 100 % amplitude, 250 kHz frequency, headstage gain ON. Then zero the signal.
- In QuadSequencer software, set up the sequence.
- Click Start and then Start again in PowerChrom software and then Run in QuadSequencer software.
How to Run a Sample
- If you haven't already done so, you should now dilute the sample (1mM LiCl, KNO3 and Na2SO4) to 100µM using deionised water.
- Check that the HVS is disarmed. Move the high electrodes out of the way and remove the BGE from reservoir 1.
- Pipette 50 µL of sample to reservoir 1 and put back the high electrodes into the reservoirs.
Notes
Preparing the Chip
Unlike glass microfluidic chips, plastic chips don’t normally require to be preconditioned before use. Researchers using plastic chips such as PMMA poly(methylmethacrylate) have reported they simply flushed the chip's channel with deionized water and then background electrolyte for a few minutes each. At the end of a working day, the microchips should be rinsed with deionized water in order to prevent the formation of salts which may clog the channels.
Positioning the Chip on the ET121 Platform
It is important that the microfluidic chip is accurately positioned on the ET121 platform. The channel inside the chip must sit directly above the golden C4D electrodes on the platform. The channel can be difficult to see with the naked eye.
One technique is to fill the channel with BGE or distilled water, and slide the chip while observing the C4D signal in PowerChrom software. Slide the chip until the max C4D signal is obtained, and then turn the two screws to secure the chip in that place
Positioning of the High Voltage Electrodes
Applying a high voltage to a solution can cause the formation of gas bubbles at the electrodes. The gas bubbles can prevent the flow of electrical current. It can also result in an electric arc which produces high temperatures and can damage the chip. You should avoid positioning the high voltage electrodes inside the hole at the very bottom of reservoir, that leads to the chip's channel. The formation of a gas bubble inside this hole could prevent the flow of current through the channel.
The high voltage can cause gas bubbles of species like OH-, H+, H2 and O2 to be produced. This can produce changes in the pH of the solution in the reservoirs, especially if a very low volume of liquid is being used. These changes in pH can cause a loss of reproducibility in the separation and detection of the analytes.
Selection of C4D Frequency and its effect on Peak Shape
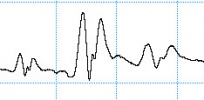
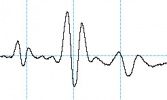
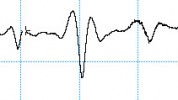
Selecting an unsuitable C4D frequency can greatly change the shape of the analyte peaks. The electropherograms below show the same analysis recorded using different C4D frequencies. When measuring potassium, sodium and lithium in acetic acid, negative peaks are expected. The first run at a C4D frequency of 1000 kHz produced what looked like three triplet peaks. When the frequency was reduced to 700 kHz, the negative peaks became more obvious. The optimum frequency was found to be at 250 kHz.
The overshoot or “shoulders” on either side of each peak has been observed and reported before.
- Pumera et al. reported (using a different microchip electrophoresis with C4D system they built) that a negative signal was observed in front of each cation peak at frequencies higher than 350 (potassium), 250 (sodium), and 200 kHz (lithium). The exact reason for the peak distortion and fluctuations observed at high frequencies is not fully understood in view of the complex nature of the registered high-frequency impedance signal. It can be attributed to the complicated equivalence circuit of the detection cell.
- Mayrhofer et al. saw overshoot or “shoulders” on either side of each peak when using C4D with capillary electrophoresis, in this research paper. The authors state this overshoot is “due to complicated vectorial sum of complex currents flowing in the spatially distributed RC network of the cell.”
Hints and Tips
- Make sure current is flowing! The cross display in QuadSequencer Software shows the current flowing through each reservoir; you should make sure there is current flowing where expected. Air bubbles in the channel can prevent the current from flowing and cause arcing which can result in high temperatures. If no current is flowing, you won't get any peaks! With 1000V across chip during injection, with acetic acid BGE in channel, you should get about 100 µA ; With 1000V applied along channel during separation, you should measure about 5-10 µA (or 10-20 µA when applying 2000V). If the voltage or current numbers are in red, there is an issue; see the QuadSequencer Software manual
- Monitor the current. A stable and reproducible current is wanted. Sudden drop or fluctuations in the measured current suggest there is a bubble or particle in the chip’s channel.
- You can increase high voltage (to 3000V for example) after the analyte peaks have been measured, to flush out the EOF more quickly. Note that this may cause Joule heating resulting in sloping baseline.
- Don’t use C4D Profiler to try to obtain best C4D settings; the C4D Profiler gives C4D settings that give heights signal, but not best peak shape
- It is sometimes necessary to filter the BGE and sample solutions to prevent particles from blocking the narrow channel of the chip. This can be achieved by using a filter which fits onto the end of the syringe.
- It may also be necessary to place the BGE and sample solutions in an ultra-sonic bath to remove any dissolved air in the solutions. Dissolved air can cause the formation of air bubbles in the channel, which can result in an electric arc when the high voltage is applied. This produces high temperatures in the channel which can damage the channel of the chip.
- Observing the current flowing through the channel of the chip can be useful when developing a method. This current is displayed in the Sequencer software, and it can be recorded from the monitor connectors at the back of the HVS. If the current reads zero, or is very noisy, during the separation step, this suggests there is an air bubble in channel.
- If the channel of the chip becomes blocked with particles, it may be possible to clear the obstruction, either by injecting air into one end of the channel using a syringe, or by using a weak vacuum at one end of the channel. Use a lint-free tissues, as opposed to normal tissues, to prevent particles from blocking the channel of the chip.
- If the sample peaks get progressively smaller over successive runs, this may be due to the depletion of ions in the sample reservoir close to the start of the channel. You can mix the sample in the reservoir, by using a pipette to suck the sample in and out of the pipette a few times.
- If the baseline begins to drift a lot during the analysis, it may be useful to flush the chip using the syringe. Place the syringe into the outlet reservoir and gently press on the syringe and hold the pressure for ten seconds.
- You may have to the alter the Offset and change the Range several times during a series of runs, but do NOT alter the Frequency, Amplitude or Headstage Gain settings until you have completed all the calibration and sample runs in an experiment.