Simultaneous Analysis of Anions and Cations in Drinking Water by CE-C4D
The successful application of C4D detection by capillary electrophoresis for the monitoring and quantification of ten inorganic ions commonly found in drinking waters (ammonium, potassium, sodium, calcium, magnesium, lithium, chloride, nitrate, sulfate and nitrite) is described [1]
Introduction
The C4D from eDAQ was used in conjunction with an Agilent G1600 capillary electrophoresis system. Design of experiments (DoE) was employed as an extensive and fast approach for method optimization (highest selectivity and sensitivity within the shortest analysis time), and as a result the studied ions were baseline separated in less then 3 minutes. The optimized analytical method was validated in terms of specificity, linearity, precision, accuracy and limits of detection and quantification. Furthermore, the method was successfully applied for the assessment of inorganic ion content of 51 geotagged drinking water (domestic well) samples and correlations between sample composition and their geographical origin were identified by multivariate data analysis.
This original research was conducted by Ioan Ovidiu Neaga, Bogdan Cezar Iacob and Ede Bodoki at the Iuliu Hatieganu University of Medicine and Pharmacy, Romania
Equipment Required
- C4D hardware unit, either:
- ER225 C4D Data System including PowerChrom software or
- ER815 C4D Detector with third-party data acquisition system
- ET120 C4D Headstage
- Agilent G1600 capillary electrophoresis system
Working Conditions
- Known sample: aqueous solution containing 15 ppm ammonium carbonate, 15 ppm potassium nitrate, 15 ppm calcium chloride, 10 ppm magnesium chloride, 10 ppm lithium sulfate, 15 ppm sodium nitrite
- Buffer: 15 mM pyromellitic acid, 10 mM citric acid, 2 mM 18-crown-6 ether adjusted at a pH of 3.70 with histidine
- CE Instrument:
- Separation voltage +30 kV
- Capillary: PVA coated fused silica capillary
- Total length 60 cm, effective length 18 cm
- Outer diameter 360 μm, internal diameter 50 μm
- Sample injection: hydrodynamic, dual opposite end injection; First the sample was injected at the inlet applying a pressure of 500 mbar, followed by the second injection at the outlet, applying a negative pressure of 50 mbar for 5 seconds at the inlet
- C4D settings: frequency 1000 kHz; amplitude 100%; headstage gain: ON
- Data recording: low-pass filter: 5 Hz; range: 50 mV; sampling rate: 1000 data points per second with the aid of Chart v.5.5.8 software
Results
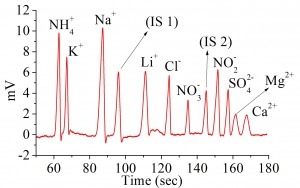
The electropherogram is shown in Figure 2. The limits of detection and limit of quantifications are shown in the table below.
- The C4D detected both anions and cations in a single injection. This was done by injecting the sample at both ends of the capillary and placing the C4D headstage at an appropriate position along the capillary.
- It was noted that C4D detection is an alternative and more flexible method of detection compared to the standard optical detection. The effective separation length can easily be adjusted by simply sliding the C4D headstage along the capillary, significantly facilitating the method development and the obtained separation efficiency.
- The estimated limits of detection (0.07 – 2 ppm) and defined limits of quantification (0.3 – 7 ppm) were comparable or better than those described for indirect UV detection, allowing much broader linear ranges (up to 120 ppm) for most of the studied ions.
| Ion | NH4+ | K+ | Na+ | Li+ | Cl- | NO3- | NO2- | SO42- | Mg2+ | Ca2+ |
|---|---|---|---|---|---|---|---|---|---|---|
| LODc (mg/L) | 1.54 | 0.45 | 0.075 | 0.094 | 2.51 | 0.92 | 0.076 | 0.901 | 0.44 | 2.33 |
| LOQc (mg/L) | 4.67 | 1.39 | 0.22 | 0.28 | 7.62 | 2.80 | 0.23 | 2.73 | 1.33 | 7.08 |
Reference
[1] The analysis of small ions with physiological implications using capillary electrophoresis with contactless conductivity detection; Ioan Ovidiu Neaga, Bogdan Cezar Iacob, Ede Bodoki; Journal of Liquid Chromatography & Related Technologies. Volume 37, Issue 14, 2014. DOI: 10.1080/10826076.2013.825862